 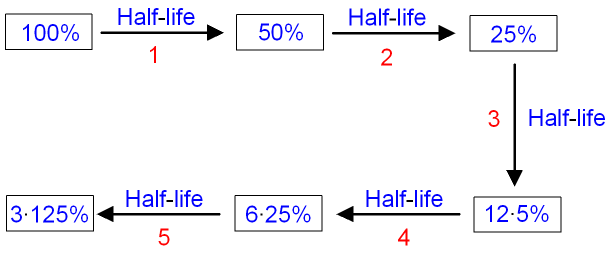
Next comes the question of how scientists use this knowledge to date things. The radiocarbon half-life or decay rate has been determined at 5,730 years. So if we started with 2 million atoms of carbon-14 in our measured quantity of carbon, then the half-life of radiocarbon will be the time it takes for half, or 1 million, of these atoms to decay. 5 It’s defined as the time it takes half a given quantity of a radioactive element to decay. The standard way of expressing the decay rate is called the half-life. Knowing the number of atoms that decayed in our sample over a month, we can calculate the radiocarbon decay rate. If we know what fraction of the carbon atoms are radioactive, we can also calculate how many radiocarbon atoms are in the lump. 4 So if we weigh a lump of carbon, we can calculate how many carbon atoms are in it. Since each beta particle represents one decayed carbon-14 atom, we know how many carbon-14 atoms decayed during that month.Ĭhemists have already determined how many atoms are in a given mass of each element, such as carbon. To measure the rate of decay, a suitable detector records the number of beta particles ejected from a measured quantity of carbon over a period of time, say a month (for illustration purposes). Different carbon-14 atoms revert to nitrogen-14 at different times, which explains why radioactive decay is considered a random process. Not all radiocarbon atoms decay at the same time. The ejected electrons are called beta particles and make up what is called beta radiation. 3 A neutron breaks down to a proton and an electron, and the electron is ejected. Carbon Dating: Determining the Rate of Radiocarbon DecayĪfter radiocarbon forms, the nuclei of the carbon-14 atoms are unstable, so over time they progressively decay back to nuclei of stable nitrogen-14. So even we humans are radioactive because of trace amounts of radiocarbon in our bodies. Radiocarbon then enters animals as they consume the plants (figure 2). Through photosynthesis carbon dioxide enters plants and algae, bringing radiocarbon into the food chain. Radioactive and non-radioactive carbon dioxide mix throughout the atmosphere, and dissolve in the oceans. This carbon dioxide, now radioactive with carbon-14, is otherwise chemically indistinguishable from the normal carbon dioxide in the atmosphere, which is slightly lighter because it contains normal carbon-12. Continued photosynthesis and feeding replaces the 14C atoms lost from the plants and animals by decay back to 14N (nitrogen-14). Radiocarbon is thus incorporated into plants by photosynthesis and into the animals that eat the plants. Radiocarbon ( 14C or carbon-14) atoms combine with oxygen atoms in the atmosphere to form carbon dioxide (CO 2) that circulates into the biosphere.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
